 The principle of liquid-liquid extraction is to create contact between the liquid mixture of constituents to separate and a solvent, and thus to generate two liquid phases in which the loaded components are distributed. 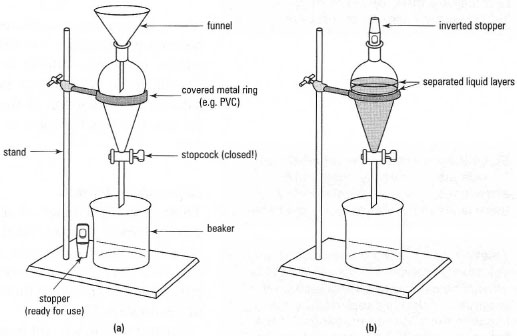
PRINCIPLE OF SINGLE-STAGE LIQUID-LIQUID EXTRACTION – PARTITION COEFFICIENT In the binary phase zone, the breakdown between the two phases follows the lever rule:.In the case of water-phenol mixtures, the CST is 66☌ and corresponds to a composition with 68% water mass. On a temperature composition diagram, points P and A progress along two curves that move closer to one another when the temperature rises ‒ until a limit where immiscibility disappears: at this temperature called “ critical solution temperature” (or CST), the solubility curves meet and points P and A are conflated.An increase in temperature generally raises the respective solubilities of two non-miscible bodies.point A represents the water-rich phase which contains a small amount of phenol.Īt this temperature of 15☌, the water’s maximum solubility in phenol is 26% mass, while the phenol’s maximum solubility in water is 8%.Point P represents the phenol-rich phase which contains a small amount of water.Point M (located between P and A) represents this mixture globally it is a fictitious point which symbolises a system comprised of two distinct phases.This is the realm of partial immiscibility. For example, if one prepares a mixture of 50% water and 50% phenol, the system separates spontaneously into two phases whose water compositions are, respectively, 26% and 92%. No compositions are possible at this temperature between P and A. 
homogeneous solutions) exist: this is the realm of total miscibility. The segments between 0% and P and between A and 100% are the ranges in which real mixtures (i.e. Immiscibility: example of a water/phenol mixture at 15☌ It also takes place over successive stages. Liquid-liquid extraction which, in the presence of a carefully chosen solvent, creates balances between two immiscible liquid phases, one of which contains a majority of the target product and the other of which contains a majority of the non-target product.Extractive distillation which, with the addition of a solvent, artificially increases the difference in volatility but requires an additional column then to distil such solvent.In such cases, other processes should be considered. However, when separating products with very similar volatility ‒ assuming a few degrees temperature difference in boiling point ‒ distillation is no longer adapted or economical. 
Separation takes place in packed or plate columns in which, plate after plate, a balance is reached successively between the steam and liquid in each of them. It is a process that relies on the difference in boiling points, namely the difference in volatility between the products to be separated. In the majority of chemicals plants, distillation is the most commonly used technique for physical separation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
